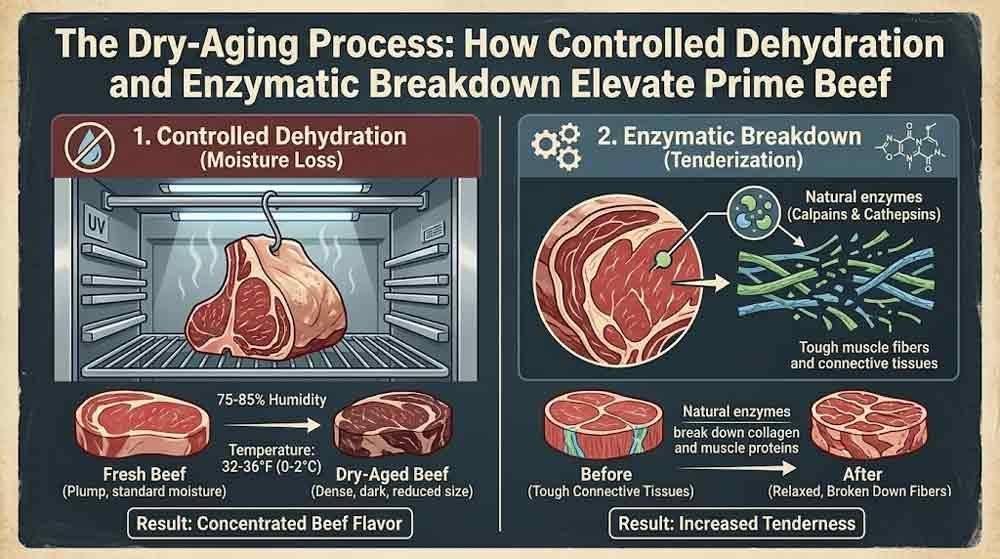
Patience is not a virtue widely celebrated in modern food culture. We live in an era of flash-frozen convenience, vacuum-sealed shortcuts, and overnight shipping of everything — including dinner. And yet, the finest steaks in the world are made slowly, deliberately, in dark refrigerated rooms where beef hangs for weeks at a time in near-silence, yielding to forces that no accelerant can replicate: time, airflow, cold, and biology. Dry-aging is one of the oldest food preparation techniques in existence, and it remains stubbornly irreplaceable — not because of nostalgia, but because the science behind it is extraordinary.
What follows is a precise account of what actually happens inside a dry-aging chamber, from the moment the carcass arrives to the moment the pellicle is trimmed and the steak hits fire.
The Beginning: What Happens to Muscle After Slaughter
To understand dry-aging, you must first understand what happens to beef in the hours immediately following slaughter. The moment oxygen is cut off from the muscle, a cascade of chemical events begins. Glycogen and glucose within the tissue break down anaerobically into lactate, causing a rapid acidification of the muscle and triggering the phenomenon known as rigor mortis — the stiffening of muscle fibers into a dense, tough, nearly inedible state. In cattle, this typically occurs within 10 to 24 hours post-slaughter (DryAger USA, 2024).
Only once rigor has fully set in does the aging process — wet or dry — become meaningful. The lactic acid produced during that initial glycogenolytic breakdown acts on the muscle fibers, and protein-cleaving enzymes called proteases begin to loosen the cellular bonds holding everything together. The meat softens. Flavor precursors accumulate. The biological clock of aging begins.
Dry-aging simply takes that natural process and extends it under precisely controlled conditions — temperature between 0°C and 4°C (32°F to 39°F), relative humidity between 75% and 85%, and consistent airflow — for anywhere from 21 days to well beyond 60. Each of those variables is non-negotiable. A deviation in any direction compromises the entire process (PMC, 2016).

Calpains, Cathepsins, and the Architecture of Tenderness
The central actors in the transformation of tough beef into something you can cut with a fork are two families of enzymes: calpains and cathepsins. Both are naturally present in muscle tissue, and both are concentrated within the meat itself — no external intervention required.
Calpains are calcium-dependent proteinases that fragment the structural proteins of muscle fiber, particularly the myofibrillar proteins responsible for the stiffness associated with rigor mortis. They work quickly and decisively in the early weeks of aging, attacking the scaffolding of the muscle at the Z-discs — the connection points between individual muscle fiber units — and essentially dismantling the architecture of toughness from the inside out (Tatnuck Meat & Sea, 2023).
Cathepsins operate more gradually. They are lysosomal enzymes — meaning they exist within specialized cellular compartments called lysosomes — and their primary role is the further breakdown of proteins into peptides and free amino acids. This is where flavor chemistry begins in earnest. The free amino acids liberated by cathepsins serve as direct flavor precursors, particularly glutamate — the compound most associated with umami, the fifth taste identified by Dr. Kikunae Ikeda in 1908. Research published in the Journal of Food Science (2025) confirms that glutamate levels in dry-aged beef rise by 30% to 45% over the first 21 days of aging, driving the deep savory intensity that defines a properly aged steak (Ribeiro et al., 2025).
A third enzyme class — lipases — completes the biochemical picture. Lipases break down intramuscular fat molecules into free fatty acids, which then undergo further oxidation into volatile aromatic compounds: the ketones, aldehydes, and alcohols that produce the signature nutty, buttery, roasted notes of well-aged beef (ScienceDirect, 2020).
The Pellicle: A Living Shield
As moisture evaporates from the surface of aging beef over the first several days, something remarkable occurs: a hard, dark exterior layer forms over the meat. This is the pellicle — often mistakenly assumed to be a sign of spoilage, when in fact it is one of the most important structural features of the entire process.
The pellicle serves multiple simultaneous functions. It acts as a physical barrier against environmental contaminants, inhibiting the growth of harmful pathogenic bacteria on the muscle surface. It slows the rate of moisture loss to a controlled pace, preventing the case-hardening effect that results from overly rapid surface drying. And crucially, it provides a stable substrate on which a consortium of beneficial microorganisms — primarily psychrophilic molds and osmophilic yeasts — can establish themselves and begin contributing their own enzymatic activity to the aging process (Environmental Literacy Council, 2025).
The balance of humidity in the aging chamber directly governs how the pellicle develops. At relative humidity below 75%, the crust forms too rapidly, potentially sealing moisture inside the muscle before adequate evaporation has occurred. At humidity above 85%, spoilage bacteria can gain a foothold. The narrow band between these extremes is where the dry-ager operates, threading a precise needle between too much and too little (ScienceDirect, 2024).

The Microbial Layer: When Mold Becomes an Ingredient
Perhaps the most counterintuitive element of dry-aging is this: beneficial mold is not an accident or a contamination. It is, in the most expert operations, a feature.
Within 7 to 10 days of aging, psychrotolerant (cold-tolerant) fungal species begin colonizing the pellicle surface. The dominant genus in most controlled dry-aging environments is Thamnidium, a mold that produces collagenolytic enzymes — enzymes capable of breaking down the perimysial connective tissue surrounding muscle fiber bundles. This enzymatic activity penetrates several millimeters into the meat below the crust, dramatically enhancing tenderness beyond what calpains and cathepsins alone can achieve (Ribeiro et al., 2025).
Mucor and Penicillium species also colonize the surface, contributing robust protease activity that liberates additional peptides and free amino acids — the Maillard reaction precursors that produce the browned, roasted flavors when the steak finally meets high heat. Concurrently, yeast species such as Debaryomyces hansenii colonize microscopic fissures in the crust, producing glutaminase and arginase that further deamidate glutamine into glutamate, amplifying umami character.
These fungi collectively form what researchers now call an enzymatic crust — a living biological system that both protects the interior meat and actively works to improve it (Ribeiro et al., 2025). The entire outer layer is trimmed and discarded before service, but the flavor compounds it drove into the underlying muscle are permanent. This is the same biological logic that governs the rind on Brie and the bloom on Roquefort. Controlled surface microflora producing flavor in the interior without spoiling it — ancient technique, modern biochemical explanation.

Concentration: The Physics of Flavor Intensity
Beyond the enzymatic and microbial chemistry, there is a simpler physical phenomenon at work: dehydration. Dry-aged beef loses between 6% and 15% of its original weight as moisture during the aging period, and in extended 45-day aged cuts, that loss can exceed 30% (Steak Locker, 2024). Every drop of water that leaves the meat concentrates the flavor compounds that remain.
This is the “angel’s share” of beef aging — a term borrowed from whiskey distillers who account for the volume of spirits that evaporates through barrel walls during maturation. The loss is real, and it is the primary reason that dry-aged beef commands a price premium at retail and in restaurants. You are paying not only for the cost of refrigeration, space, and time — you are paying for what has been removed. The resulting concentration of beefy, savory volatiles is chemically measurable. Research using UPLC-MS metabolomics has confirmed that dry-aged samples contain significantly greater concentrations of flavor-active free amino acids, nucleotides, and volatile organic compounds compared to wet-aged counterparts — particularly inosine monophosphate (IMP) and its umami-synergizing interaction with glutamate (ScienceDirect, 2020).
There is also a textural consequence to dehydration that manifests at the moment of cooking. Fresh beef contains a high percentage of surface moisture that must evaporate before the Maillard reaction — the high-heat browning reaction responsible for seared crust development — can begin. Dry-aged beef has already lost that surface moisture during aging, meaning that when it contacts a scorching cast-iron surface or a live fire grill, the Maillard reaction initiates instantly and proceeds with extraordinary intensity. The crust forms faster, darker, and with more complexity than on any fresh-cut steak (Kasapi Blog, 2025).
Duration and the Flavor Curve: When Does Aging Become Overaging?
The relationship between aging duration and flavor development is not linear. The tenderness gains from enzymatic breakdown are most dramatic in the first 21 to 28 days, leveling off substantially after the five-week mark as the primary calpain activity diminishes. Flavor complexity, however, continues to evolve well past that point.
Research on grass-fed beef loins documents that tryptophan — one of the free amino acids liberated by cathepsin activity — peaks around Day 21 at approximately 0.8 mg/g before converting into indolic compounds like 3-indoxyl sulfate, which contribute subtle bitter and earthy aromatic notes (Ribeiro et al., 2025). Glycine and alanine accumulate steadily throughout aging, imparting sweet and briny undertones. At 40 days and beyond — particularly in highly marbled breeds such as Wagyu or Hanwoo — the synergistic interaction between glutamate and IMP reaches maximum intensity, producing the kokumi flavor profile that Japanese food scientists describe as a richness and depth that extends the perception of umami on the palate (ScienceDirect, 2020).
Most premium steakhouses operate in the 28-to-45-day window, balancing tenderness optimization with flavor development and commercial yield practicality. Extended aging beyond 60 days is the domain of specialists — producers and chefs willing to absorb the shrinkage losses in pursuit of a flavor profile that is, by any objective measure, among the most complex naturally occurring in cooked food.
Safety, Regulation, and the Misconception of Risk
A persistent hesitation among consumers unfamiliar with dry-aging is the assumption that aged beef must be dangerous — that the visible mold and darkened exterior represent spoilage. The science does not support this concern when the process is properly managed.
Current risk assessments from the European Food Safety Authority (EFSA) and major international regulatory bodies confirm that, when time-temperature parameters are respected — specifically storage between -0.5°C and 3.0°C with relative humidity between 75% and 85% and consistent airflow of 0.2 to 0.5 meters per second — dry-aged beef carries no greater microbiological risk than fresh meat (Ribeiro et al., 2025). Enterobacteriaceae are suppressed in the interior muscle throughout the process. Foodborne pathogens, when detected at all in metagenomic analysis of 90-day aged samples, appear at levels below actionable thresholds.
The mold species most commonly colonizing the pellicle — Thamnidium, Mucor, and Penicillium — rarely produce mycotoxins under standard dry-aging conditions, and the crust that contains them is trimmed entirely before cooking. What reaches the plate is the interior muscle: microbiologically clean, biochemically transformed, and organoleptically extraordinary.
The Irreplaceable Logic of Slow
Dry-aging is, in the end, an argument against acceleration. Every shortcut that food science has devised — electric stimulation to speed post-slaughter tenderness development, enzyme injection, accelerated wet-aging at elevated temperatures, even experimental 48-hour aging using Aspergillus oryzae — produces a product that trained palates consistently rank below the result of genuine slow aging. The complexity of flavor generated by weeks of concurrent enzymatic breakdown, microbial succession, moisture concentration, and lipid oxidation cannot be compressed into a shorter timeline without losing something essential.
A 45-day dry-aged ribeye is not merely a more tender steak. It is a biochemically different object than the fresh-cut version from the same animal — its protein architecture dismantled and reassembled into something softer, its flavor compounds multiplied and intensified, its surface primed for the most complete Maillard reaction possible, its microbiome having worked in cooperative succession to produce an enzymatic crust that flavored the interior from the outside in.
Time, it turns out, is not a luxury in the production of exceptional beef. It is the primary ingredient.
Sources
- Ribeiro, D.M., et al. “Dry-Aged Beef: A Global Review of Meat Quality Traits, Microbiome Dynamics, Safety, and Sustainable Strategies.” Journal of Food Science, Wiley Online Library, 2025. https://ift.onlinelibrary.wiley.com/doi/10.1111/1750-3841.70589
- “The Science of Dry Aging.” DryAger USA. https://www.dryagerusa.com/blogs/know-how/the-science-of-dry-aging
- “The Science Behind Dry-Aging.” SteakAger, October 2024. https://steakager.com/blog/the-science-behind-dry-aging
- “How Does Dry-Aged Beef Not Spoil?” Tatnuck Meat & Sea, 2023. https://tatnuckmeatandsea.com/fresh-meat/beef/how-does-dry-aged-beef-not-spoil/
- Kim, Y.H.B., et al. “Elucidating Mechanisms Involved in Flavor Generation of Dry-Aged Beef Loins Using Metabolomics Approach.” Food Research International, ScienceDirect, 2020. https://www.sciencedirect.com/science/article/abs/pii/S0963996920309947
- “Effects of Relative Humidity on Dry-Aged Beef Quality.” Meat Science, ScienceDirect, 2024. https://www.sciencedirect.com/science/article/abs/pii/S0309174024000755
- Dashdorj, D., et al. “Dry Aging of Beef: Review.” PMC/National Library of Medicine, 2016. https://pmc.ncbi.nlm.nih.gov/articles/PMC4872334/
- “Characterizing the Flavor Precursors and Liberation Mechanisms of Various Dry-Aging Methods.” PMC, 2022. https://pmc.ncbi.nlm.nih.gov/articles/PMC9230681/
- “How Is Meat Dry Aged Without Going Bad?” Environmental Literacy Council, 2025. https://enviroliteracy.org/how-is-meat-dry-aged-without-going-bad/
- “The Science Behind Dry Aging.” Kasapi Blog, January 2025. https://www.kasapi.bg/blog-post-1-en.html
- “What Does Dry Aging Do to a Steak’s Flavor?” Steak Locker. https://www.steaklocker.com/blogs/blog/what-does-dry-aging-do-to-a-steak